
Clove oil is an essential oil that can be derived into various aroma chemicals for flavor and fragrance ingredients.1 The main components in clove oil are eugenol and caryophyllene, which could be synthesized into more than 15 derivatives such as isoeugenol, methyl isoeugenol, and vanillin.1 The latest synthesis technology uses sustainable innovation and green chemistry, such as a catalytic process for isoeugenol and dihydroeugenol, which have 100% atom economy, meaning all reagents are converted into the desired product without producing waste that pollutes the environment. The same thing happens in developing catalytic oxidation technology for the synthesis of vanillin made from eugenol, using green chemistry resulting in environmentally friendly and economical processes.
 Leo Seno Broto
Leo Seno Broto
Sources for Vanillin Production
Vanillin is an essential flavoring component widely used in food and beverages and the flavor and fragrance industries. The current trend for naturals has led to the rapid increase in demand for natural vanillin globally.
Vanillin can be produced from numerous sources such as petroleum origin (guaiacol), spruce tree lignin (coniferyl alcohol), corn sugar (glucose), rice bran (ferulic acid), vanilla beans (vanillin glucoside) and clove oil (eugenol).2
Natural Vanillin ex Clove Authentication
Most of the vanillin in the market comes from chemical synthesis, with approximately 85% produced from guaiacol (petrochemical products), while the rest are from lignin and eugenol. The difference in the starting raw materials for vanillin production affects the organoleptic profile, molecular fingerprint, regulatory product status, and the price of the resulting products. For example, vanillin from eugenol has a different sensory profile than vanillin from guaiacol and lignin. Vanillin ex clove is considered natural ingredient in the United States, while vanillin from guaiacol and lignin are considered as synthetic. Vanillin is also produced from ferulic acid to answer growing market for EU natural vanillin.3
The difference in the price range of vanillin from the various raw materials mentioned above merits a reliable authentication method that can accurately determine the “authenticity” of the vanillin source, namely the use of Stable Isotope Ratio Analysis–Isotope Ratio Mass Spectrometry (SIRA-IRMS) and Site-Specific Natural Isotopic Fractionation by Nuclear Magnetic Resonance (SNIF-NMR).3,4
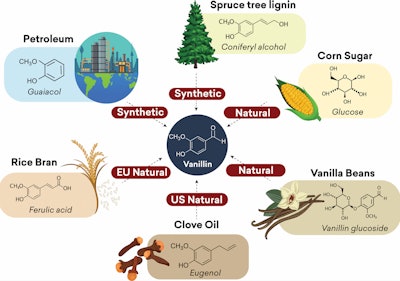 F-1. Various sources of vanillin starting raw materials.
F-1. Various sources of vanillin starting raw materials.
SIRA-IRMS is a method used to determine the authenticity of a molecule by measuring the abundance of stable isotope ratios for 13C/12C, 18O/16O, and 2H/1H of an element in a molecule that is specific to its geographic origin, type of plant photosynthesis, source of raw material, and the process path of synthesis of vanillin molecules. The origin of the vanillin raw material can be measured by the deviation value of the carbon ratio 13 (d13C), which is the value between the ratio of ratio 13C/12C sample to 13C/12C standard and calculated using the formula d13C = [(13C/12C) sample/(13C/12C) standard) – 1 ] x 1000.3, F-2 shows the results of IRMS measurements for Vanillin from various sources of raw materials.5
Isotope Ratio Mass Spectrometry analysis showed Vanillin authentication based on the ratio of stable isotope 13C/12C of various precursors such as petroleum and plants in various photosynthesis pathways (Calvin synthesis (C3 plants), Hatch-Slack pathway (C4 plants), or Crassulacean acid metabolic (CAM)), with different values as shown in figures 2.4,5 As seen in F-2, the d 13C range of values for vanillin ex clove is in the range of -32 – (-) 30, while synthetic vanillin from the guaiacol figure is in the range of -36 – (-)34, semi-synthetic vanillin from lignin falls in the range of -28.5 – 26.5.5.
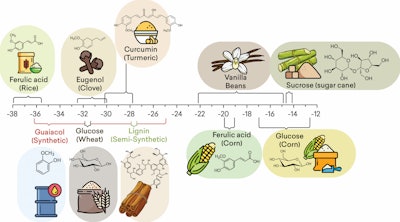 F-2. Delta 13C range of values for vanillin according to a specific source and the chemical used as a precursor by SIRA IR
F-2. Delta 13C range of values for vanillin according to a specific source and the chemical used as a precursor by SIRA IR
The addition of delta 2H isotope analysis on top of the d 13C as shown in F-3, aims to further improve the accuracy of vanillin authentication. One of the disadvantages of only using the IRMS d 13C method is that it can be manipulated by the addition of cheaper materials that have an abundance of C-13 isotopes. This addition will cause the deviation value of carbon isotopes to shift positively toward natural vanillin from vanilla bean or the lower range of d 13C of vanillin sourced from other raw materials.5,6
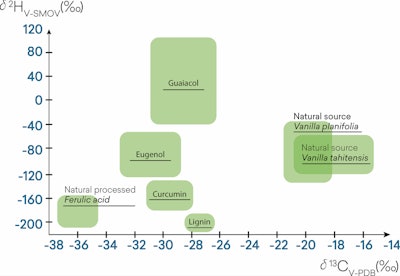 F-3. Multielement IRMS range of value depending on the source of vanillin4
F-3. Multielement IRMS range of value depending on the source of vanillin4
The most accurate method in Vanillin authentication is SNIF-NMR, an analysis method that quantitatively measures the Nuclear Magnetic Resonance of deuterium (2H NMR) and carbon-13 (13C NMR). This method will give an abundance value that is specific to each element, namely eight carbon positions and five hydrogen positions in the Vanillin molecule, which depends on the origin of the vanillin precursor material, the plant origin of the Vanillin source, and the route of the Vanillin synthesis process used.5,6 The ability of SNIF-NMR to determine the source of vanillin is shown in F-4, namely from the principle component analysis (PCA) graph. This method can clearly show the origin of the raw material for the same molecule, the geographical origin of the molecule, and the composition of the vanillin mixture. Sources of vanillin raw materials such as eugenol, guaiacol, lignin, ferulic acid from rice bran, ferulic acid from corn, and vanillin mixture made from the guaiacol-ferulic acid mixture can be distinguished significantly.6
Combination of the two analytical methods, SIRA-IRMS and SNIF NMR, accurately show the origin of the raw materials to the level of distinguishing the botanical sources used and the composition of natural and synthetic vanillin mixing, if any. The use of authentication technology is critical in guaranteeing the authenticity status of vanillin from fraud for consumers and economic sustainability for producers who entirely use the material in compliance with the regulatory status of vanillin.
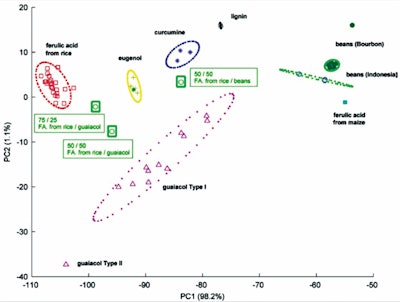 F-4. Two-dimensional plot of the scores obtained by PCA on the values of the 13C isotopic composition for all eight sites obtained by SNIF-NMR
F-4. Two-dimensional plot of the scores obtained by PCA on the values of the 13C isotopic composition for all eight sites obtained by SNIF-NMR
Conclusion
Vanillin is a crucial material that is widely used in different applications. Assuring that the vanillin purchased is according to the regulatory claim made by the producer could be checked using these specific methods. It is of utmost importance to make sure of the authenticity of vanillin, such as vanillin ex. clove. This prevents further adulteration or mixing of synthetic vanillin into natural vanillin.
References
1. Razafimamonjison G., Jahiel M., Duclos T., Ramanoelina P., , Fawbush F., and Danthu P. 2014. Bud, leaf, and stem essential oil composition of Syzygium aromaticum from Madagascar, Indonesia and Zanzibar. International Journal of Basic and Applied Sciences, 3 (3) (2014) 224-233.
2. Bomgardner M. The problem with Vanilla: After vowing to go natural, food brands face a shortage of the favored flavor. Chemical and Engineering News. Accessed 1 September 2022, https://cen.acs.org/articles/94/i36/problem-vanilla.html
3. Desmurs, J.R, Giannotta D., Gelo-Pujic M., Role C., Lancelin P. Synthesis and Autentication of Natural Vanillins Prepared by Fermentation. Techniques, Regulations, source Variations and Flavor Characteristics. Rhodia Perfumery. Perfumer and Flavorist. Allured Publ. 29, (1), 32–41.
4. Christoph N., Antje Schellenberg, Wiebke Zander, Gerhard Krammer. 2017. Stable Isotope Ratio Analysis for Authenticity Control. Springer Handbook of Odor, 2017. ISBN : 978-3-319-26930-6
5. Pironti C., Ricciardi M., Motta O., Camin F., Bontempo L., and Proto. Application of 13C Quantitative NMR Spectroscopy to Isotopic Analyses for Vanillin Authentication Source. Foods 2021, 10, 2635. https://doi.org/10.3390/foods10112635
6. Guyader S., Thomas F., Jamin,E., Grand M., Akoka S., Silvestre V., and Gérald S. 2019. Combination of 13C and 2H SNIF-NMR isotopic fingerprints of Vanillin to control its precursors. Flavor and Fragrance Journal. 20 January 2019. https://doi.org/10.1002/ffj.3486











